Background
Cold atmospheric plasma (CAP) is a partially ionized gas generated by electrical discharges at room temperature and atmospheric pressure. CAP is highly reactive: it produces radiation in the form of UV and visible light, free electrons and electric fields (EFs), and reactive oxygen and nitrogen species (RONS). Over the past decade, CAP has emerged as a promising therapeutic tool for wound healing, with RONS considered key mediators of its biological effect.
Wound healing is among the most complex processes in the human body, involving a tightly coordinated yet overlapping sequence of hemostasis, inflammation, growth, re-epithelialization, and remodeling of the extracellular matrix (ECM). Successful wound repair first requires immune cells to clear the wound of damaged skin cells and bacteria to prevent infection. The tissue growth phase that ensues involves fibroblasts and keratinocytes migrating toward the wound to rebuild tissue, and the remodeling phase involves ECM reorganization by fibroblasts to generate a resistant elastic scar. Studies using animal models show that CAP enhances several of these steps: it stimulates ECM secretion, promotes angiogenesis, and reduces the bacterial load of the wound. Clinical trials using CAP have also shown encouraging results, including reduced bacterial load and faster healing. However, we still lack full understanding of which components of CAP are responsible for the observed effects of CAP on wound healing.
In their recent study, “Cold atmospheric plasma promotes migration persistence, through induced H2O2 and electric field,” published in the February 3, 2026 issue of Biophysical Journal, Labérie et al. address this gap. This exciting new study investigates the individual and combined effect of H₂O₂ and EFs on the migration persistence of single fibroblasts, revealing an additional critical role of EFs in CAP-induced migration.
The setup
In this study, Labérie et al. turned to a simple but physiologically relevant model, primary human fibroblasts isolated from skin. The authors exposed cells to a large range of doses of CAP by using a homemade single-electrode setup for the plasma jet. This setup allows precise control over the delivered doses, which ranged from 0.37 up to 5 J.mL-1, by varying exposure time while keeping power constant. To reconstruct individual trajectories, cells were recorded and tracked by videomicroscopy by using phase contrast optics. Cell tracks were analyzed by calculating migration persistence, speed, and mean-square displacement (MSD). By disentangling persistence from speed, their analysis could determine whether CAP makes cells move faster or steers them more directionally.
Key findings
Effects of CAP alone
Using MSD analysis to track single fibroblasts revealed that, at low CAP doses, cells explored a larger area and followed straighter trajectories. This behavior was due to increased migration persistence, which refers to a directional memory or measure of how long a cell maintains directional migration before turning. Thus, CAP enhances directional memory of migration in fibroblasts rather than just speed.
In addition to being dose dependent, the effect of CAP was transient. The increase in migration persistence was more pronounced during the first 8 h after treatment, weakened at 8–16 h, and absent by 24 h. Interestingly, adult fibroblasts displayed a weaker response than neonatal fibroblasts, prompting Labérie and colleagues to focus on neonatal fibroblasts for the rest of their study.
Lamellipodia and Arp2/3: a structural link to CAP-induced migration persistence
Labérie and colleagues examined whether the effect of CAP on persistence was mediated by the Arp2/3 complex, known to regulate this process in other cell systems. The Arp2/3 complex is responsible for nucleating branched actin networks at the leading edge of migrating cells. When inhibiting Arp2/3 with the small molecule CK-666, not only was baseline persistence reduced, but the CAP-induced increase in directional migration was completely abolished. Thus, Arp2/3-dependent lamellipodial protrusion acts as the cytoskeletal (structural) link through which CAP promotes persistent migration.
Dissecting CAP-generated signals: reactive oxygen species and EFs
Having established that CAP enhances migration persistence through actin remodeling, the authors focused on which CAP-generated components could be mediating this behavior. First, they focused on hydrogen peroxide (H₂O₂), the best characterized reactive oxygen species implicated in CAP-mediated wound healing. When fibroblasts were treated with H₂O₂ alone, low concentrations had little effect, but increasing the dose to just 30 μM was sufficient to significantly increase migration persistence. In contrast, treatment with other CAP-generated RONS (NO2− and NO3–) did not affect migration persistence.
The authors then investigated transient EFs, which are also generated by CAP, but whose contribution remains largely unexplored. To isolate this component, the authors engineered plasma setups that reduced the EF strength while maintaining similar levels of reactive species. This setup enabled them to observe how reducing field strength caused the CAP-induced increase in persistence to disappear. Instead, when applied independently of plasma, EFs modestly enhanced directional migration.
Together, these findings indicate that both H₂O₂ and EFs can independently modulate fibroblast migratory behavior; yet, neither is sufficient on its own. This led the authors to postulate that CAP may act through their combined action. To test this, they combined the two stimuli to mimic CAP conditions and found that low-dose H₂O₂ paired with an externally applied EF was able to recapitulate CAP-induced increase in persistence. The same synergy was also observed at the structural level, with increased lamellipodial width and greater percentage of cells displaying lamellipodia. Collectively, these findings indicate that CAP promotes migration persistence through the combined action of chemical (H₂O₂) and physical (EF) cues that converge on the actin cytoskeleton.
Conclusions
This work examines the role of CAP on the migration of human primary dermal fibroblasts (Figure 1). By analyzing single-cell trajectories, Labérie and colleagues find that CAP exposure enhances directional persistence. Interestingly, a short 10-s exposure is sufficient to induce a migratory effect in neonatal fibroblasts that lasts for up to 16 h, revealing CAP hysteresis. Mechanistically, CAP treatment appears to initiate an intrinsic self-sustained process driven by Arp2/3-mediated lamellipodia, which commits cells to a persistent migration mode via positive feedback loops that involve branched actin and the small GTPase Rac1.
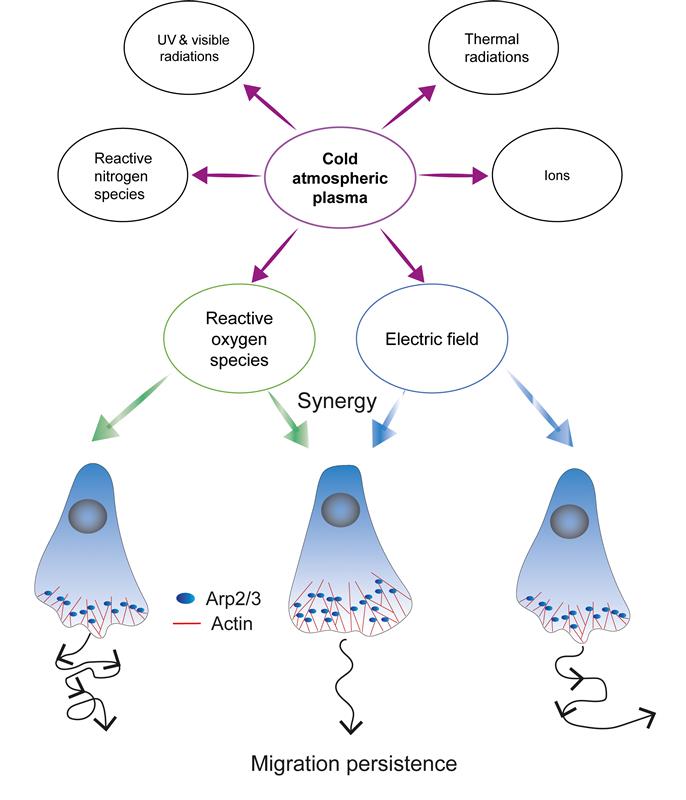
Figure 1: CAP-induced migration persistence. Schematic illustrating CAP-generated products, highlighting reactive oxygen species and transient EFs as the two key signals that synergistically converge on Arp2/3-dependent lamellipodia to drive fibroblast migration persistence. Figure taken from Labérie et al. 2026.
The authors dig deeper by identifying the specific CAP-generated signals mediating this behavior. Rather than acting through a single stimulus, CAP shapes cell behavior via the synergistic action of two cues: H2O2 and transient EFs. These two signals converge on actin-driven lamellipodia to promote migratory persistence. Although each stimulus alone produces only modest changes, together they fully recapitulate persistent fibroblast migration induced by CAP.
Taken together, these novel findings not only help explain the effectiveness of CAP in wound healing therapies but also highlight a broader concept: combining chemical and electrical cues can represent a powerful tool to direct cell behavior during tissue repair.